GP referral
Value Based Procurement Pilot Releases Theatre Time and Inpatient Bed Capacity
The ability to carry out UROLIFT outside of the main hospital complex has meant that we were able to resume this service much earlier than our in-patient elective work during the COVID-19 pandemic. We now have no significant waiting list for UROLIFT, unlike for many of our other non-urgent procedures for which waiting lists are extremely long, as is the case across the NHS. This has meant that, at least for the proportion of men with bladder outlet obstruction who are suitable for UROLIFT, we have been able to treat this very common urological condition and give symptomatic relief to many men, who otherwise would still be waiting for their surgery.
Malcolm Crundwell, Consultant Urologist, Royal Devon and Exeter NHS Foundation Trust
The Challenge
Royal Devon and Exeter NHS Foundation Trust provides acute and community services to people in Exeter, East Devon and Mid-Devon. In total the trust has around 845 beds and employs around 11,000 staff. The population served is approximately 460,000 which increases significantly in peak summer holiday season.
There was an urgent need to free up theatre capacity and inpatient beds. Along with other acute trusts in the region, Royal Devon and Exeter struggled to meet waiting time targets, with some patients waiting over a year for non-cancer surgery.
The standard surgical treatment offered to men with lower urinary tract symptoms (LUTS) from benign prostatic obstruction (BPO) is transurethral resection of the prostate (TURP). Before the COVID-19 pandemic, the trust was performing around 150 TURP operations each year, with an average inpatient stay of three days. TURP is performed under a general anaesthetic, taking an average of 90 minutes of theatre time. Patients normally have an indwelling catheter for a few days following a TURP and some are discharged with the catheter in situ, requiring a return to clinic for a trial without catheter.
The Aim
- Create a minimally invasive day case treatment pathway for men needing surgery for BPO.
- Free up theatre and inpatient bed capacity in the main acute hospital.
The Solution
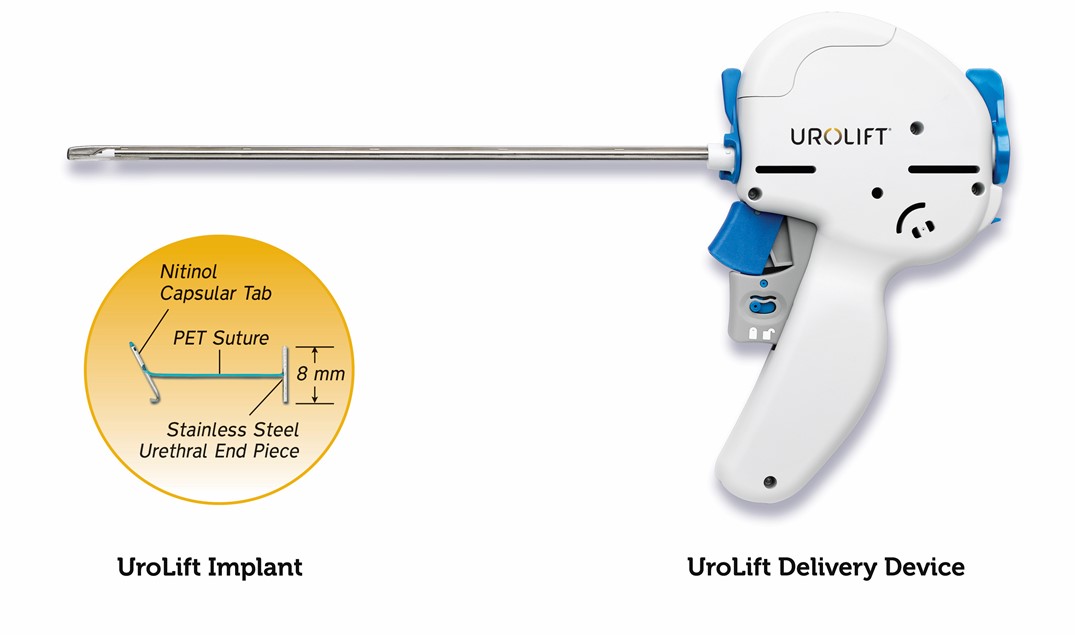
The UROLIFT System is a short, minimally invasive procedure that can be performed under a local anaesthetic. Completed in a day surgery unit or an outpatient setting, with patients discharged within a few hours, typically without a catheter.
The system uses adjustable, permanent implants to retract the prostatic lobes and reduce urethral obstruction so that it does not narrow or block the urethra. In this way, the device is designed to relieve symptoms of urinary outflow obstruction without cutting or removing tissue.
The National Institute for Health and Care Excellence (NICE) recently updated its guidance on UROLIFT, stating that the evidence supports the case for adopting the system for treating LUTS of BPO. It should be considered as an alternative to transurethral resection of the prostate (TURP) and holmium laser enucleation of the prostate and can be done as a day-case or outpatient procedure.
The Process
Eligible patients were recruited from the urology waiting list or from outpatient clinics. UROLIFT lists were being scheduled once to twice each month. Six patients were routinely treated in a single half-day morning theatre session. Patients are typically discharged in the afternoon following the procedure in the morning and followed up in clinic at six weeks. Typically, patients are discharged without a catheter.
The first cohort of 93 patients treated with the system from July 2017 to May 2018 were audited and the results published as a NICE Shared Learning Case Study. The key benefits and changes to pathway are shown below.
The second cohort of 110 patients from April 2019 to March 2020 were audited as part of the Value Based Procurement project. This case study compares the resource utility and cost of UROLIFT.
The Results
The UROLIFT System enabled the trust to move the BPO system out of its main hospital, Royal Devon and Exeter, and introduce the system to the community hospitals in Tiverton and Exmouth.
Transforming the patient pathway in this way:
- Reducing length of stay. This is a day case procedure compared with TURP which is associated with an average inpatient bed stay of three days.
- Reducing theatre time. UROLIFT is a short procedure under local anaesthetic compared with TURP, which is a 90 minute procedure and requires a general anaesthetic.
- Relieves pressure on the main hospital beds and theatres.
- The financial benefit driven from this initiative was used by the trust to help mitigate the cost impact of growth in numbers of procedures.
TURP
Acute Hospital LUTS assessment
Management plan discussed with patient
TURP surgery – 90 minutes (average) under general anaesthetic
Inpatient stay – 3 days (average)
Discharge with or without catheter
Outpatient app to remove catheter – Trial without catheter (TWOC)
Outpatient follow up at 6 weeks
UroLift
GP referral
Acute Hospital LUTS assessment
Management plan discussed with patient
UroLift procedure – 15-20 minutes (average) under local anaesthetic
Discharge typically without catheter
Telephone or outpatient follow up at 6 weeks
Financial Benefits
When the trust started doing Urolifts there was a requirement for additional consumable’s funding however the change in procedure reduced bed days and theatre time for this cohort of patients. Further to this the work was carried out in a community theatre creating much needed capacity in main theatres and more effective use of the community site.
Rachel Cobby, Senior Directorate Management Accountant, Surgical Services Division
Local financial analysis was carried out for this case study. This confirmed that, whilst the higher cost of the Urolift consumables compared with TURP these are offset by the savings in inpatient bed stay and theatre time, when carried out as a day case procedure.
Based on the experience at Royal Devon and Exeter, which, in 2019-20, treated 110 patients with UROLIFT as an alternative to TURP:
- Average reduction per case of 60 mins of theatre time and 2.6 inpatient bed days.
- Annual release of 110 hours of theatre time at a cost of £84,546.
- Annual release of 290 inpatient bed days at a cost of £123,076.
- Average saving per patient of £424 when UROLIFT performed as day case, creating a total net annual saving of £46,686.
In addition to the above NICE have provided guidance and an economic assessment in relation to this product. However, it should be noted that the benefits demonstrated in the evaluation by NICE are based on the assessment undertaken by the Economic Assessment Centre. Providers would need to undertake their own assessment based on their own costs and the capacity benefits that would be achieved by implementing these technologies.
Operational Benefits
- Enabled treatment of patients in a community day case unit away from the main hospital. This has freed up capacity for more urgent work such as cancer treatment.
- Improved patient experience. No serious complications or serious side-effects.
- Reduced burden on waiting times.
Further Opportunities
The trust is considering how to move the procedure into an outpatient or ambulatory setting, which will create further efficiencies and cost savings.
A Urology Investigation Unit (UIU) is now open in the Ottery St Mary community hospital. The plan is to develop a “one-stop” service for investigation of LUTS in men. This will comprise history and examination, flow rate and bladder residual volume, flexible cystoscopy and/or transrectal ultrasound as appropriate. The one-stop service will also provide the opportunity to discuss treatment options, with the patient either deciding on the day or having the opportunity to reflect on their options. The patient will then return on a separate date for his procedure.
For those men electing for UROLIFT, the aim is to do this in the UIU under local anaesthetic (LA). This one-stop service will allow assessment of the suitability of men for UROLIFT under a LA. It will ensure that those men who would not tolerate a LA, will still be able to have the procedure done under sedation in an operating theatre.
Next Steps
This project highlights the potential of transitioning from inpatient to day case and the benefits that can be achieved. There is growing support too from NICE, British Association Of Day Surgery (BADS) and Getting It Right First Time (GIRFT) for performing UROLIFT in an ambulatory setting, which could be either in the acute setting or in the community. Employing UROLIFT at scale to implement this type of pathway transformation for patients with BPO across an ICS footprint could release significant capacity and resource.
A review of services for bladder outlet obstruction across the regional network in Devon and Somerset is being carried out. The aim is to produce a more coordinated approach to this very common condition. Leading to the ability to offer men a full range of treatment options with seamless transfer from one unit to another, as appropriate.
See our Useful Links section for more information on how Value Based Procurement can support your organisation and key contacts.
Please note: This case study was carried out at Royal Devon and Exeter NHS Trust in conjunction with Teleflex. There may be other comparable solutions within the market that are suitable in addressing the challenges highlighted in this report.
UROLIFT System FAQs
Teleflex provides a dedicated clinical team, providing clinical training and support. It is also able to arrange clinical proctoring where needed. For further information please contact 01494 532 761.
Yes. The OPCS procedure code for UROLIFT (M68.3; Endoscopic insertion of prosthesis to compress lobe of prostate) maps to HRG LB70 (Complex Endoscopic, Prostate or Bladder Neck Procedures).
Links section
-
Value Based Procurement
How we are working with the NHS to shift from cash releasing savings to value based procurement.
-
VBP Contacts
Find contact information for our Value Based Procurement Leads for the Category areas.
-
Collaborative Working Reduces Overnight Stays For Parotid Surgeries
Successful Pilot reduces overnight stays for parotid surgeries at Manchester NHS Foundation Trust.
-
Innovative Technology Reduces Operating And Recovery Time
Successful Pilot Reduces Operating and Recovery Time At Guy's And St Thomas' NHS Foundation Trust.
-
Getting It Right First Time
National Day Surgery Delivery Pack.
-
The MedTech Funding Mandate
The NHS Long Term Plan committed to accelerate the uptake of selected innovative medical devices, diagnostics and digital products.
